|
Practice applying your understanding by playing the 3rd and 4th game levels. Mass Number- the total number of protons and neutrons in a neutrons Atomic Number- The number of protons in the nucleus of an atom, which determines the chemical properties of anĮlement and its place in the periodic tableĭ. Charge- the electric- The electric charge is delivered from the formula: p + e.Ĭ. Element Symbol- The number of protons in the element determines the element the element symbol is 1,2 or 3 lettersī. Create a definition (using a complete sentence) for each of these items based on your labels from the atomic symbol above.Ī.Position in symbol box Term to describe thisinformation Particle used to determinethis How the value isdeterminedĪ Element symbol protons # of p will identify theelement (Label each of the letters below as to their significance and what they stand for.) Using the Symbol readout box, figure out which particles affect each component of the atomic symbol and how the value of.Write a rule for determining the mass number of an atom or ion? proton + neutron = mass numberġ0 applying your understanding by playing 1st and 2nd levels on the game screen. Play with the simulation to discover what affects the mass number of your atom or ion. There is a positive charge if there are fewer electrons there is a negative charge if there are more electrons Negative ions have fewer protons than electrons.ĭevelop a relationship (in the form of a single sentence or equation) that can predict the charge based on the number and Positive ions have more protons than electrons.Ĭ. Neutral atoms have the same number of protons and electrons.ī. electronsįill in the blanks below to show your results:Ī. Play with the simulation to discover which particles affect the charge of an atom or ion. An atom with 4 protons and 4 neutrons: beryllium An atom with 2 protons and 4 neutrons: heliumĬ. An atom with 3 protons and 4 neutrons:lithiumī. 
Play until you discover which particle(s) determine(s) the name of the element you build._Ī. What particle(s) are found in the center of the atom? protons and neutrons _ you can make different elements with different proton and neutron combinationsĬlick on the + sign for each of the boxes (element name, net charge and mass number) to view changes as you change the you get ions when you add protons to neutronsī. (Put all answers in either BLUE ink or with HIGHLIGHTER)Ī. As you explore, talk about what you find. Explore the Build an Atom simulation with your group. Simulation (lorado/en/simulation/build-an-atom)Ģ. Click on HTML5 simulations on top right of screen and choose the Build an Atom Can you ever have more than this number in the first energy level? Can you ever have less? 4.Name _ Partner Name _ Date _ Period _īuilding an Atom (PhET simulation) PART I: ATOM SCREEN 1. How many electrons can fit in the first energy level? 3. The first energy level is the one closest to the nucleus, and the second is furthest away. The model shows two orbits, which we often call energy levels. Where are the electrons located inside of the atom? Make sure that the simulation is set to the “orbital” model. When you are ready to start the exercise, press the “Reset All” button in the bottom left corner to clear any changes.An ion means there is an overall charge to the atom. Note that when particles are added to your model you can see if the atom is stable or unstable (labeled in the center), and if it is neutral or an ion (labeled on the outer rings).The “Element”, “Mass” and “Net Charge” boxes should be expanded. You can add particles to your atom by dragging them into the atom model. Play around with the simulation for a bit to become familiarize with the controls and functions.Click the green “Run Now” button below the large image to start the application. 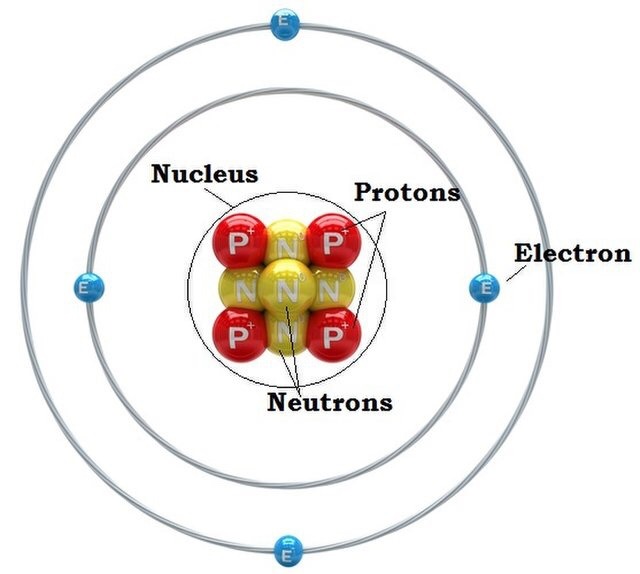
(Round the atomic mass from the periodic table to the nearest whole number to get part of your answer.) Directions: Describe which particles and how many of each you need to make a neutral beryllium atom. What 2 sub-atomic particles have charges? List the particle name and its charge. Predict how changing the number protons and electrons will change the element and its charge. Use information about the number of protons, neutrons, and electrons to identify an element, its position on the periodic table. 
Chemistry I Name: _ Hour: _ Date: _ “Build An Atom” Simulation – Build an Ion Learning Goals: 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
